 Brackets surround this structure, and there is a superscripted negative sign. The 100 free and reliable online calculators that help you to solve any calculation-related problems and provides you with the. The left structure shows an oxygen atom with three lone pairs of electrons single bonded to a nitrogen atom with one lone pair of electrons that is double bonded to an oxygen with two lone pairs of electrons. In terms of formal charge, a structure generally contributes more when (1) the formal charges on the atoms are minimized and (2) any negative formal charges are on more electronegative atoms and any positive charges are on more electropositive atoms. This chemistry video tutorial provides a basic introduction into how to calculate the formal charge of an atom or element in a lewis structure. 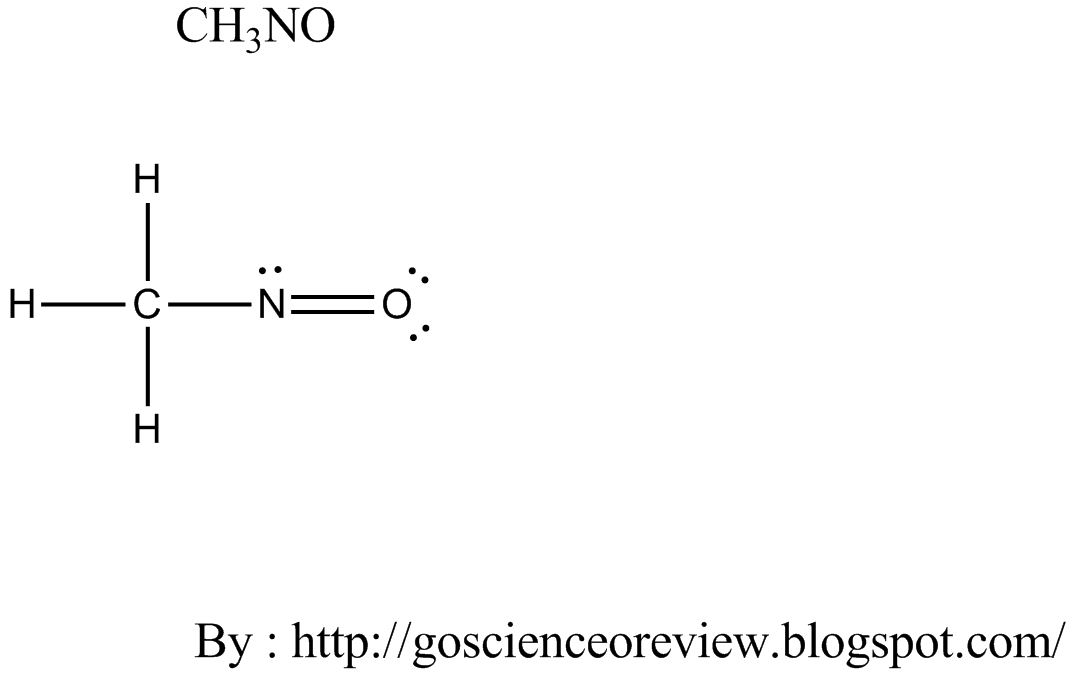
The electrons involved in the N–O double bond, however, are in different positions: When a molecule has nonequivalent resonance structures, one structure may contribute more to the resonance hybrid than another. So, for as long as you have a complete Lewis structure and periodic table handy, you can quickly find the formal charge of any atom in a molecule. 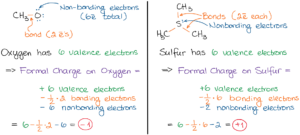 
In order to use the formula charge formula, we must observe the Lewis Dot Structure for. \) can have two possible structures with the atoms in the same positions. Mathematical Method Formal charge equation is based on the comparing the number of electrons in the individual atom with that in the structure. Step 1: Draw the Lewis Dot Structure for the compound given in order to calculate the formal charge. Struggling with Formal Charges Chad breaks down a simple way to remember the formula for calculating Formal Charge (Normal Valence minus 'dots and lines. Formal charge equation formally compares the number of valence electrons in an isolated neutral atom (which can be determined from the older style group number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
